About the ROADMAP Project
With around 30 million people in the U.S. affected by rare diseases, and approximately 7,000 of them lacking an FDA-approved therapy, drug repurposing has become a promising option to identify effective treatments faster and more affordably. However, challenges exist in this space, including a lack of consensus on stakeholder roles, the absence of a roadmap for pursuing drug repurposing, and insufficient support throughout the process.
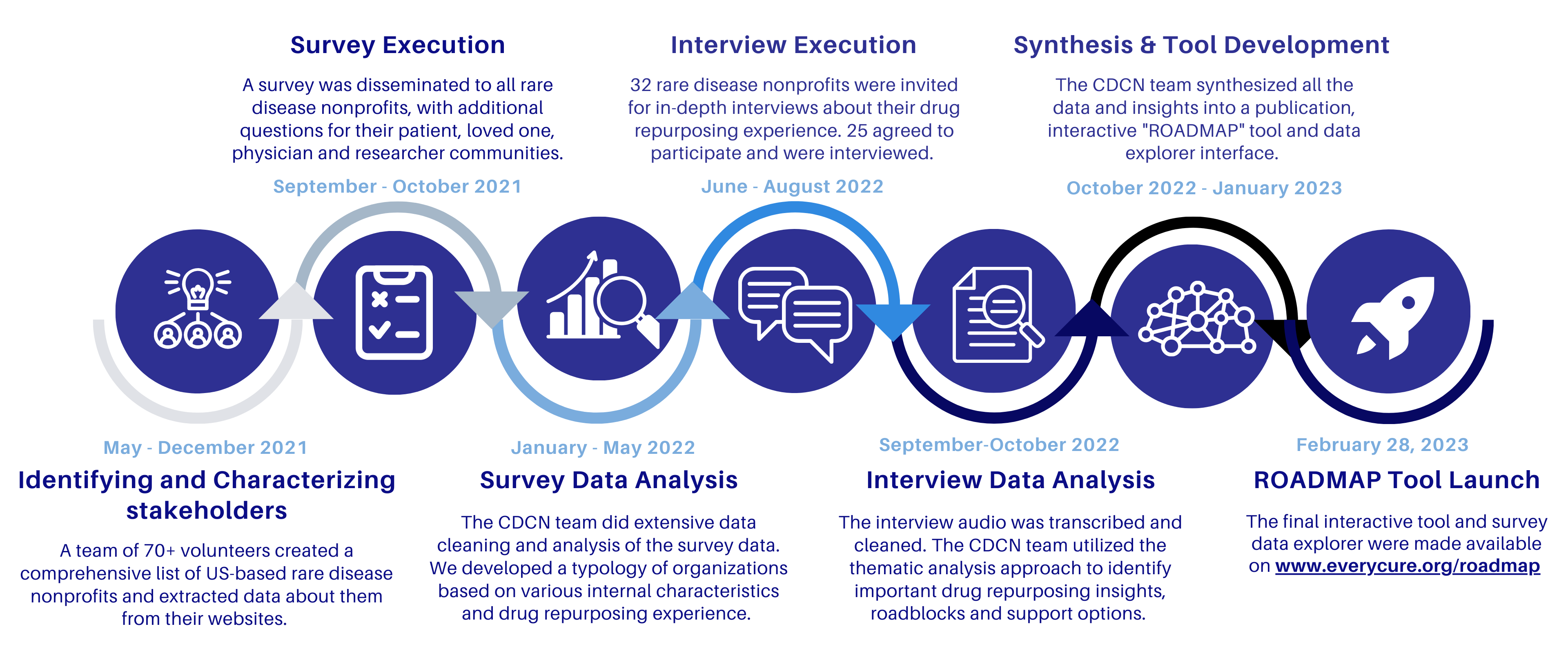
To address these challenges, the CDCN has partnered with the Chan Zuckerberg Initiative and EveryCure on the Repurposing Of All Drugs, Mapping All Paths (ROADMAP) project. The goal was to create a resource that would provide guidance and support to rare disease organizations seeking to pursue drug repurposing, based on real world experiences. Over a two-year period, the CDCN project team collected vital data and gained insights from a wide range of US-based rare disease nonprofit organizations and other their patient, loved one, physician and researcher populations through a survey and in-depth interviews, synthesizing them into an interactive tool format.
The ROADMAP tool serves as a comprehensive guide for rare disease organizations pursuing drug repurposing, offering a step by step walkthrough of the different stages and roadblocks associated with them. In each stage, we provide a comprehensive menu of options that are available, to help support informed decision-making. The tool is interactive, allowing users to navigate the various stages of drug repurposing and explore the data. We hope this new resource will provide crucial guidance and support for all rare disease organizations working to find and repurpose existing treatments!
WE WANT TO HEAR FROM YOU!
We want to hear about your experience with the ROADMAP tool! Your feedback is valuable to us as we work to improve the tool and make it as user-friendly and accurate as possible. Together, we can continue to advance drug repurposing efforts for rare diseases. Please send any thoughts, comments or suggestions to info@castlemannetwork.org.
PDF DOWNLOADS
For your convenience, we also are providing the following in PDF form if you have trouble accessing the information on the interactive tool:
For a brief overview of some of the initial insights from the ROADMAP, watch the below video to see project lead Ania Korsunska present at a meeting of the Chan Zuckerberg Rare As One organizations:
As a part of the ROADMAp, we executed a survey, which included sections for several different stakeholder types: (1) rare disease nonprofit organization leaders, (2) rare disease patients, (3) rare disease patients’ loved one (parent, spouse, friend, sibling, etc. of a rare disease patient), (4) physicians who treat rare diseases, (5) rare disease researchers.
ROADMAP Participating Organizations:

OUR TEAM
Dr. David Fajgenbaum, Ania Korsunska, Mileva Repasky, and Mary Zuccato.
And prior CDCN/CSTL team members Johnson Khor, Dr. Ruth-Anne Langan Pai, Hemi Park, and Trae Boyd
Our Super Star Volunteers (past and present, in order of onboarding)
Meg Zuccato, Anna Nguyen, Derek Ansel, Martin Lukac, Annalise Jear, Mitav Nayak, Panchatapa Baul, Samatha Hood, Sabina Grigorian, Jacob Lowy, Justin Wong, Sydney Grisham, Yuan (Abby) Feng, Leanna Chen, Megan Shieh, Bryan Aguilar, Marcy Spiker, Rose Weathers, Katherine Fang, Veikko Toikka, Robert Parillo, Lindsay McBride, Sara Barrett, Robert Parillo, Dallas Ryan, Veikko Toikka, Benita Balogun, Robert Parillo, Penny Deremer, Miti Patel, Emma Roemer, Neda Pazuki, Erikka Chowdhury, Stephanie Hage, Matt Scott, Geetha Turlapati, Jessica Xiang, Jada Watkins, Jazmin Loughlin, Justin Crawmer, Ferzana Niazi, Anaheit Arathoon, Michael Zhang, Vee Suresh, Mahima Sangtani, Sara Cronin, Rita Aberbach, Kayleigh Nicole Murray, Diane Baynes, Andrew Zhu, Anastasia Kakurina, Susanna Hunanyan, Jade Bondy, Simarsukh Dhillon, Angela Perry, Carolyn Canterbury, Owen Yu and Kanan Lathia.
For this project, the CDCN was awarded a grant from the Chan Zuckerberg Initiative, whose science program supports the science and technology that will make it possible to cure, prevent, or manage all diseases by the end of the century.